Abstract
The emerging outbreak of the coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) continues to spread worldwide. We prescribed some promising medication to our patients with mild to moderate pneumonia due to SARS-CoV-2, however such drugs as chloroquine, hydrossichloroquine, azithromycin, antivirals (lopinavir/ritonavir, darunavir/cobicistat) and immunomodulating agents (steroids, tocilizumab) were not confirmed as effective against SARS-CoV2. We, therefore, started to use auto-hemotherapy treated with an oxygen/ozone (O2/O3) gaseous mixture as adjuvant therapy. In Udine University Hospital (Italy) we performed a case-control study involving hospitalized adult patients with confirmed COVID-19 with mild to moderate pneumonia. Clinical presentations are based upon clinical phenotypes identified by the Italian Society of Emergency and Urgency Medicine (SIMEU-Società Italiana di Medicina di Emergenza-Urgenza) and patients that met criteria of phenotypes 2 to 4 were treated with best available therapy (BAT), with or without O3-autohemotherapy. 60 patients were enrolled in the study: 30 patients treated with BAT and O2/O3 mixture, as adjuvant therapy and 30 controls treated with BAT only. In the group treated with O3-autohemotherapy plus BAT, patients were younger but with more severe clinical phenotypes. A decrease of SIMEU clinical phenotypes was observed (2.70 ± 0.67 vs. 2.35 ± 0.88, p = 0.002) in all patients during hospitalization but this clinical improvement was statistically significant only in O3-treated patients (2.87 ± 0.78 vs. 2.27 ± 0.83, p < 0.001), differently to the control group (2.53 ± 0.51 vs. 2.43 ± 0.93, p = 0.522). No adverse events were observed associated with the application of O2/O3 gaseous mixture. O2/O3 therapy as adjuvant therapy could be useful in mild to moderate pneumonia due to SARS-CoV-2. Randomized prospective study is ongoing [Clinical Trials.gov ID: Z7C2CA5837].
Keywords: Autotransfusion; COVID-19; Cytokine release syndrome (CRS); Lipid ozonation products; Medical O3; O3 gas; O3 therapy; Oxidative stress; Reactive oxidative species; SARS-CoV-2.
Introduction
The emerging outbreak of the coronavirus disease 2019 (COVID-19) caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) continues to spread worldwide. According to the COVID-19 situation report published by the World Health Organization (WHO), a total of 14.3483858 confirmed cases and 603.691 deaths have been identified globally up to July 20th 2020 [1].
The full spectrum of COVID-19 ranges from mild, self-limiting respiratory tract illness to severe progressive pneumonia, multi-organ failure, and death [2].
There is a reasonable hypothesis that after a first viral phase with low severity illness, there could be a host immune response with variable lymphopenia, and an inflammatory cytokines release syndrome (CRS) which would play an important role in severe COVID-19 cases, leading to worsening acute respiratory failure [3].
In this context of CRS, patients could take advantage of O3 gas anti-inflammatory and immune-modulatory properties. On this basis, we performed a case–control study in patients with COVID-19 to assess O3-autohemotherapy as adjuvant therapy, in addition to the prescription of standard treatments used such as hydrossi-chloroquine, azithromycin, antivirals (lopinavir/ritonavir, darunavir/cobicistat) and immunomodulating agents (steroids, tocilizumab) [4, 5].
O2/O3 mixture, a gas discovered in the mid-nineteenth century, is a molecule consisting of three atoms of oxygen in a dynamically unstable structure due to the presence of mesomeric states [6].
Although O3 gas has dangerous effects when inhaled, some reports on O3 therapeutic effects have been documented.
Since the First World War, blood ozonization has shown an effective bacterial effect. Furthermore, the diffusion of several studies regarding patient infected by Ebola and treated with O3 therapy demonstrate its capacity to stimulate oxygen metabolism and to modulate the immune system. During the First World War (1914–1918), doctors familiar with O3 antibacterial properties, and with few other medical resources available, applied it topically to infected wounds, thus revealing O3 hemodynamic and anti-inflammatory properties [6,7,8].
O3 therapy administration depends on treatment goals and location of therapy: it has been administered intravenously, intramuscular, into body cavities (rectum, bladder, and vagina), joints, subcutaneous and soft tissues, intraperitoneally, and even intrathecally [9]. However, the first and most popular application is O3 autohemotherapy (O3-AHT) which requires a predetermined blood sample (100–200 mL) to be drown and thus, using stoichiometric calculations, a precise concentration of O2-O3 can be infused. This small amount of blood is subjected to O2-O3 ex-vivo and is then administered to the patients [10].
O3 has many further demonstrated mechanisms of action:
- (1)Increase of red blood cell 2,3 di-glycerophosphate (greater hemoglobin oxygen release);
- (2)Modulation of endothelial nitric oxide production;
- (3)Induction of a greater arterial/venous partial pressure difference, indicating greater mitochondrial consumption of oxygen (energy production);
- (4)Modulation of the immune system and reduction of inflammation,
- (5)Improvement of antioxidant status, antioxidant enzymes (including superoxide dismutase), and glutathione in cells [9];
In vitro O3 interferes with virus-to-cell contact in lipid-enveloped viruses via oxidation of lipoproteins, proteins, and glycoproteins, thus interfering with the viral reproductive cycles [10].
O3 in the blood reacts with plasma antioxidant and gives rise to several messengers, among them H2O2, that readily diffuses into immune cells. Moreover, O3 acts as a regulator in signal transduction, facilitating several immune responses [11, 12].
Another important effect of O3-AHT in the COVID-19 therapeutical context is the down-regulation action of pyrin domain-containing protein 3 (NLRP3) inflammasome, which is known to play a crucial part in the initiation and continuance of inflammation in various diseases [13].
The rationale of blood ozonization is to evaluate the additive anti-inflammatory and immunomodulatory action mediated by O3 in patients hospitalized for SARS-CoV-2.
Methods
Study patients
Male and non-pregnant female patients 18 years of age or older were eligible if they had a diagnostic specimen that was positive to RT-PCR for SARS-CoV2 and pneumonia confirmed by chest imaging. All patients gave their informed consent to participate in the study. Exclusion criteria were pregnancy, G6PHD (glucose 6 phosphate dehydrogenase) deficiency, concomitant serious diseases and failure to obtain informed consent.
This study was approved by the Friuli Venezia Giulia ethics committee (Protocol n° Z7C2CA5837).
The full spectrum of COVID-19 ranges from mild, self-limiting respiratory tract illness to severe progressive pneumonia, multi-organ failure, and death. The Italian Society of Emergency and Urgency Medicine (SIMEU—Società Italiana di Medicina di Emergenza-Urgenza) suggests to classify the COVID-19 patients in 5 clinical phenotypes [14]:
- Phenotype 1: subjects with fever and without respiratory failure (normal Arterial Blood Gas analysis—ABG-, six-minute walking test—6mWT—and Chest XR). These patients can usually manage at home maintaining a quarantine period.
- Phenotype 2: subjects with fever but with ABG and/or Chest XR indicative of modest respiratory insufficiency (PO2 > 60 mmHg in ambient air) and/or pulmonary consolidation area. These patients need to be hospitalized because they can worsen quickly.
- Phenotype 3: subjects with fever associated with moderate-severe respiratory insufficiency (at triage PO2 < 60 mmHg in ambient air) and/or bilateral pulmonary consolidation area at Chest XR. These patients need to be treated with high flow oxygen therapy.
- Phenotype 4: subjects with respiratory failure with suspected ARDS (Adult Respiratory Distress Syndrome) or complicated pneumonia. These patients require hospitalization in a sub-intensive care unit.
- Phenotype 5: subjects suffering from ARDS initially. These patients will require Intensive Care Unit (ICU) admission and non-invasive positive pressure ventilation (NIPPV) or mechanical ventilation.
In the present study, we enrolled subjects with modest to moderate respiratory insufficiency (SIMEU clinical phenotypes 2–4) hospitalized in an infectious disease ward.
For all patients were recorded age, gender, presence of obesity, Charlson comorbidity index, delta time from symptoms onset to hospital admission, hospital length of stay, and symptoms (fever, dry cough, dyspnea, syncope, myalgia, asthenia, diarrhea, hypo/dysgeusia, hypo/anosmia).
The other best available therapy (BAT) administered included: antiretroviral therapy (lopinavir/ritonavir 2 tablets every 12 h, darunavir/cobicistat 1 tablet per day), hidrossycloroquine 400 mg every 12 h than the first day, followed by 200 mg every 12 h for other 4 days, tocilizumab iv 10 mg/kg in single dose, amiodarone, azithromycin, other antibiotics and steroids.
The endpoints of the study were: SIMEU clinical phenotypes at admission and discharge, delta classes SIMEU (negative values mean clinical improvements), admission to Intensive Care Unit (ICU) and death. Blood gas analysis (BGA) parameters were recorded at admission. Furthermore, baseline value of White Blood Cells (WBC), lymphocytes count, platelets, CPK, LDH and IL-6 blood level were reported.
Medical ozone (O3) procedure
The systemic O3 treatment procedure was decided by the physician on duty and started first at the patient’s bedside with the drawing of autologous blood and, after proper O2/O3 mixing, with its re-infusion. After inserting a catheter in the patient’s vein which was connected to a latex-free plastic bag, containing 35 ml of sodium citrate, a blood amount of 200 ml was drown from the patient. At the end, the withdrawal line was washed and re-used to infuse saline solution. The blood, without any patient disconnection, was then mixed with a gas mixture of a 200 ml composed by 96% O2 and 4% O3, with a O3 therapeutic range of 40 μg/mL of gas per mL blood. To guarantee the O2/O3 homogeneous diffusion into the blood, the bag was gently mixed for about 10 min, subsequently, the blood was re-infused into the patient. O3 treatment lasted for 3 consecutive days.
Outcome
The end-point was the time of respiratory improvement and earlier weaning from oxygen support: these parameters were included in the SIMEU clinical phenotypes classification.
Statistical analyses
All variables were reported as mean ± standard deviation, median and interquartile interval or proportion, depending on their distribution. Accordingly, comparisons between treated and controls were performed with independent samples two-tailed t test, Mann–Whitney test or chi-squared test with continuity correction. Mixed ANOVA was used to compare the SIMEU class values, cross-classified by treatment and time (before and after treatment). Paired sample’s t test was used when appropriate. For analyses, the R statistical software was used [15].
Results
A total of 60 patients were enrolled in the study: 30 with O3-AHT as adjuvant therapy added to BAT and 30 controls treated with BAT only. Among the 30 patients treated with O3-AHT (cases group), 28 received three consecutive O3-AHT sections, and 2 received two consecutive doses.
Table 1 shows the baseline demographic and clinical characteristics of the 60 patients enrolled in the study. The control group treated with BAT only were older and with more co-morbidities than the other group. Instead, patients treated with O3-AHT as adjuvant therapy had a more serious clinical respiratory condition with lower PaO2/FiO2 and SpO2/FiO2 ratio and lower lymphocytes count.
Notably, SIMEU clinical phenotypes at hospital admission were slightly higher in O3-treated patients, meaning that their conditions were more severe, even if the difference was not fully statistically significant (2.87 ± 0.78 vs. 2.53 ± 0.51, p = 0.054).
In general, a decrease of SIMEU clinical phenotypes was observed (2.70 ± 0.67 vs. 2.35 ± 0.88, p = 0.002) in overall population during hospitalization. However, this clinical improvement was statistically significant only in O3-treated patients (2.87 ± 0.78 vs. 2.27 ± 0.83, p < 0.001) and not in the control group (2.53 ± 0.51 vs. 2.43 ± 0.93, p = 0.522).
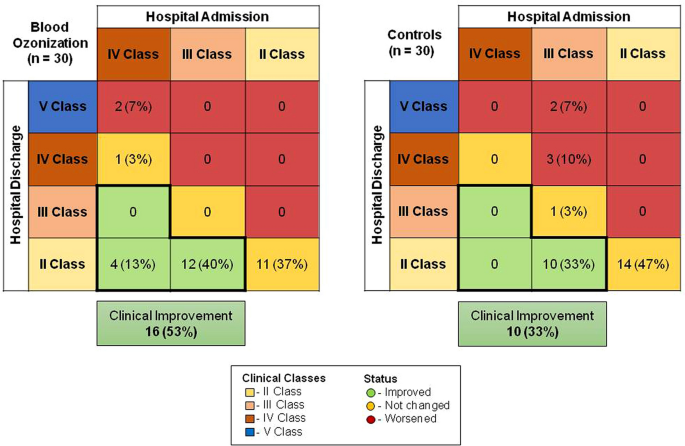
Figure 1 shows the clinical improvement (in green) in the two groups, in O3 group there was a 53% improvement with respect to 33% of the control group. In the group with O3-AHT adjuvant therapy, only 7% of patients had a worse outcome, compared with 17% in the control group.
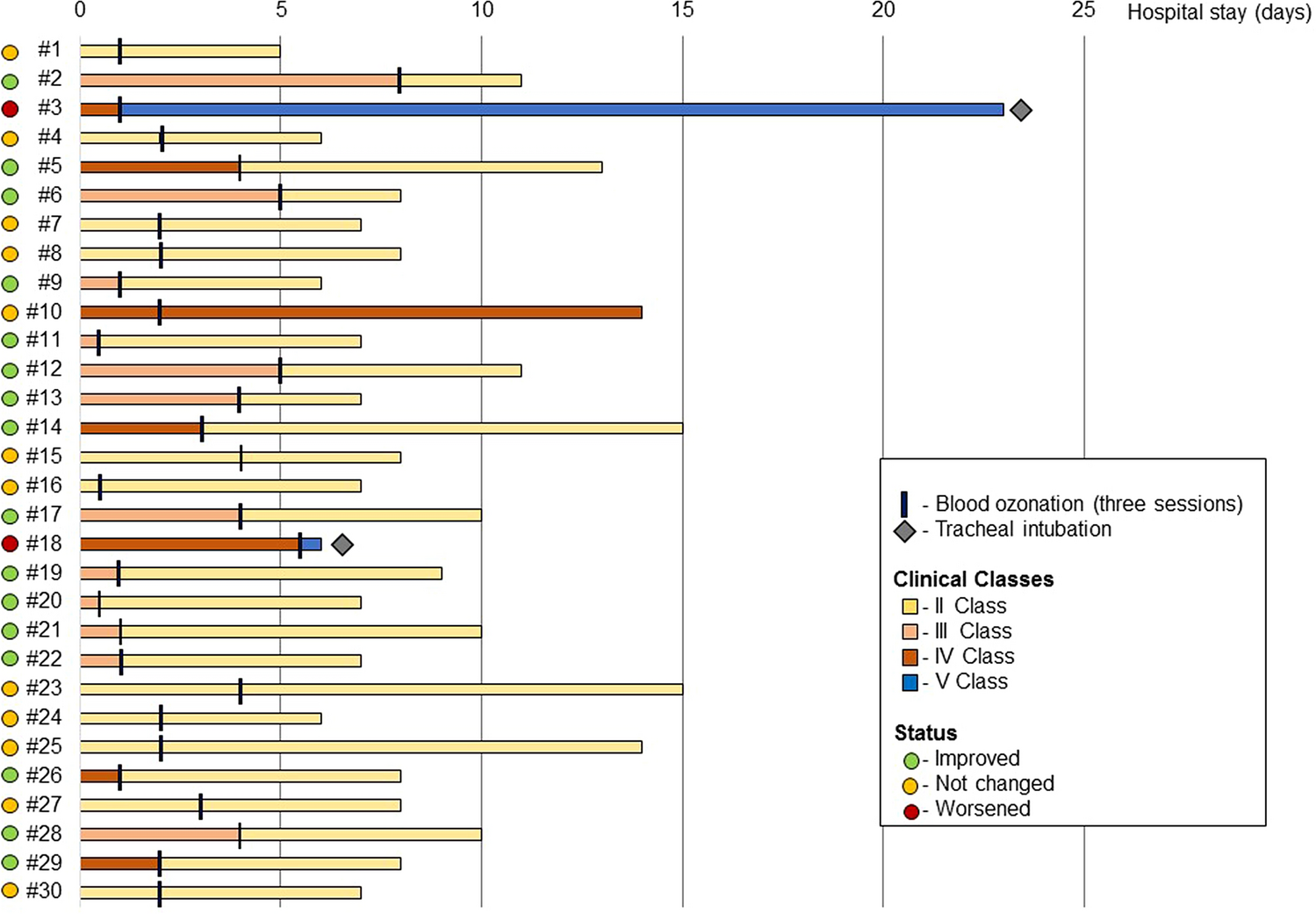
Figure 2 shows all the 30 patients treated with O3-AHT adjuvant therapy, according to the improvement of SIMEU clinical phenotypes and the time of O3-AHT therapy; 26 out of 30 patients were treated in the first 5 days after hospital admission. Four patients (two for each group) were intubated, and only two patients died in the control group.
Given the nature of this preliminary study, data on a limited number of laboratory measures were collected. No adverse events were recorded in both patients’ arms.
Discussion
This preliminary report describes the clinical outcomes in a small group of patients with COVID-19 who were moderately ill (SIMEU clinical phenotypes 2–4) and were treated with BAT plus O3-AHT with respect to BAT alone. Specifically, clinical improvement status was observed in 53% of the patients in the O3 group and 33% in the control group; instead, the clinical stability was observed in 40% of O3-AHT group and in 50% in the control group, failure was observed in 7% of O3-AHT group with respect to 17% of the control group. Mortality was observed in only two cases in the control group: the low rate of mortality may be due to the patients’ profiles enrolled in the study with mild or moderate cases of pneumonia due to SARS-CoV-2.
Case fatality Rate (CFR) in Friuli Venezia Giulia was 0.3% at July 20th 2020 while in Italy CFR was 14.3% [16], however, mortality was very different across the regions and wards of admission [17,18,19]. This mortality rate in our patients with mild to moderate pneumonia was not surprising.
None of the patients enrolled in the study were intubated at the moment of the inclusion, whereas at the end four patients were intubated, 2 for each group. In a recent study of compassionate use of remdesivir [20], 64% of patients were intubated at the baseline and mortality was 18%, instead in less severe cases, mortality was 5%, very similar to the value observed in our study.
To note that, respect to the World Health Organization guidance [21], that is based on clinical data and radiological data only for patients with ARDS and designed for clinicians caring COVID-19 patients during all phases of their disease (i.e. screening to discharge), the SIMEU clinical phenotypes [14] use both clinical and radiological criteria for all patients, allowing a better patients stratification during the first phase of the disease, especially in the field of emergency medicine and might be more precise in patients stratification during the follow-up. This classification might be more useful for evaluating the clinical and radiological improvement of patients included in a clinical trial on COVID-19.
Although several limitations, the present study suggests that the clinical effectiveness of O2/O3 therapy in addition to the standard of care ensured at Udine University Hospital in patients with mild to moderate COVID-19 pneumonia. The O3-AHT therapy is based on oxidative preconditioning [22] that leads to the reduction of IL-6 and IL-1b [23]. This effect can explain a cytoprotective action observed during O3-AHT therapy. Furthermore, O3-AHT therapy has been used in viral infection (Ebola, Influenza and HIV) with preliminary good results [10, 24].
Unfortunately, our study did not collect viral load data to confirm the anti-viral effects of O2/O3 therapy.
No adverse events were detected during O3-AHT therapy, neither haemolysis or local vein damages.
Interpretation of the results of this study is limited because of the small size of the enrolled group, the short duration of follow-up, the potential missing data due to the retrospective nature of this study, and the lack of a randomization.
Although the latter precludes definitive conclusions, comparisons with contemporaneous cohorts from the literature suggest that O2/O3 therapy may have a clinical benefit in patients with mild to moderate COVID-19 pneumonia.
Physicians desperately need therapy for COVID-19. Published case reports might support inexpensive O2/O3 utility for infectious disease management, not only for microbicidal activity but also for the immunomodulation activity. O3 (and other oxidation) therapy carries virtually no known adverse or toxic effects when performed properly (other than sporadic vein issues, as can other intravenous therapies).
Me (C.T.) has never administered O3-AHT therapy before COVID-19, and the discussion with the anaesthesiologist (A.D.M.) brought us to try a therapy with few adverse effects for an infection without any available therapy. O2/O3 is not the panacea for all diseases, however, evidence-based medicine strongly suggests that randomized prospective studies are necessary. On this basis, according to the ethical committee of Region Friuli Venezia Giulia, Italy, we started a multicentre prospective randomized study of therapy with BAT with or without O3-AHT adjuvant therapy for mild to moderate pneumonia due to COVID-19. The study is currently ongoing [Clinical Trials.gov ID: Z7C2CA5837].
Descarcati articolul Tascini2020_Article_BloodOzonizationInPatientsWith



2 Comments
sikis izle
I am always looking online for ideas that can facilitate me. Perl Trefor Bondy
cbd gummies for sleep and anxiety
I like it whenever people get together and share opinions.
Great website, keep it up!